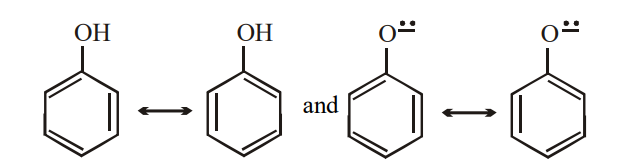
A) General reaction pathway for hydrogenation of phenol. (B) Possible... | Download Scientific Diagram
Phenol hydrodeoxygenation: effect of support and Re promoter on the reactivity of Co catalysts - Catalysis Science & Technology (RSC Publishing) DOI:10.1039/C6CY01038E
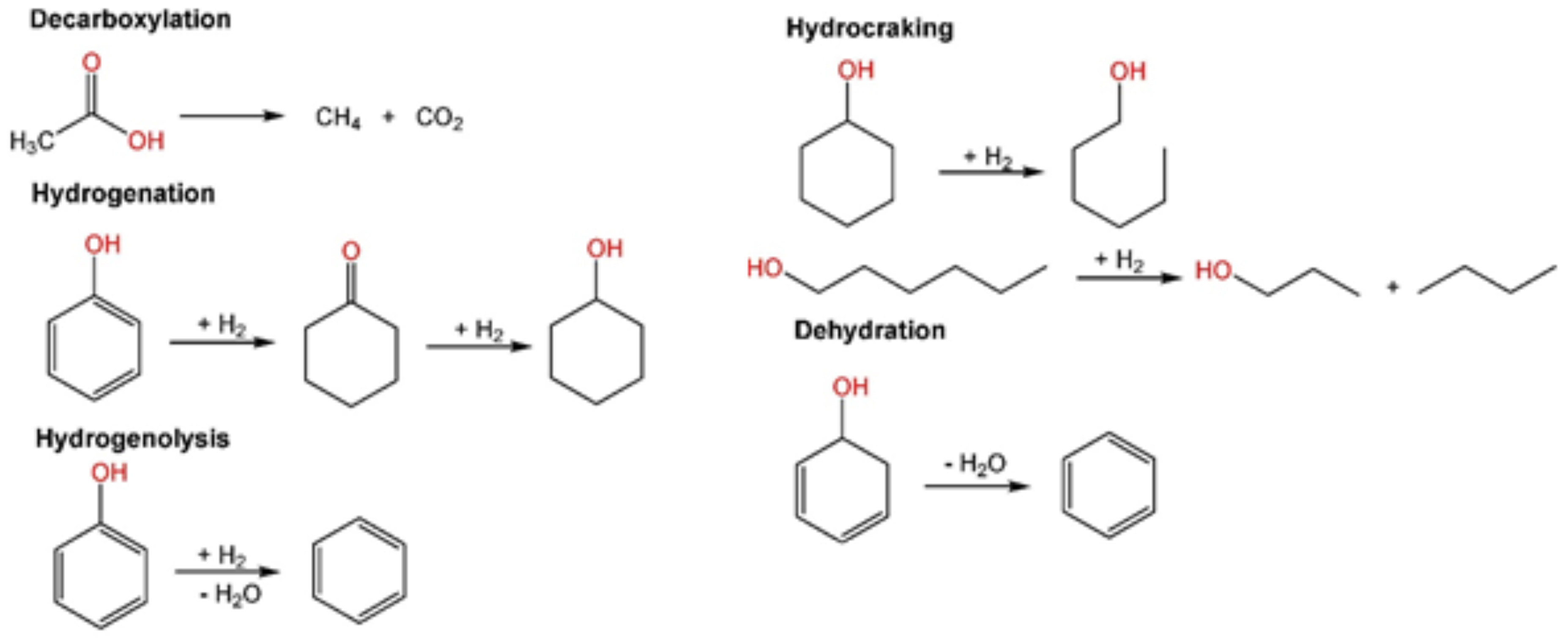
Energies | Free Full-Text | Fast Pyrolysis Oil Upgrading via HDO with Fe-Promoted Nb2O5-Supported Pd-Based Catalysts
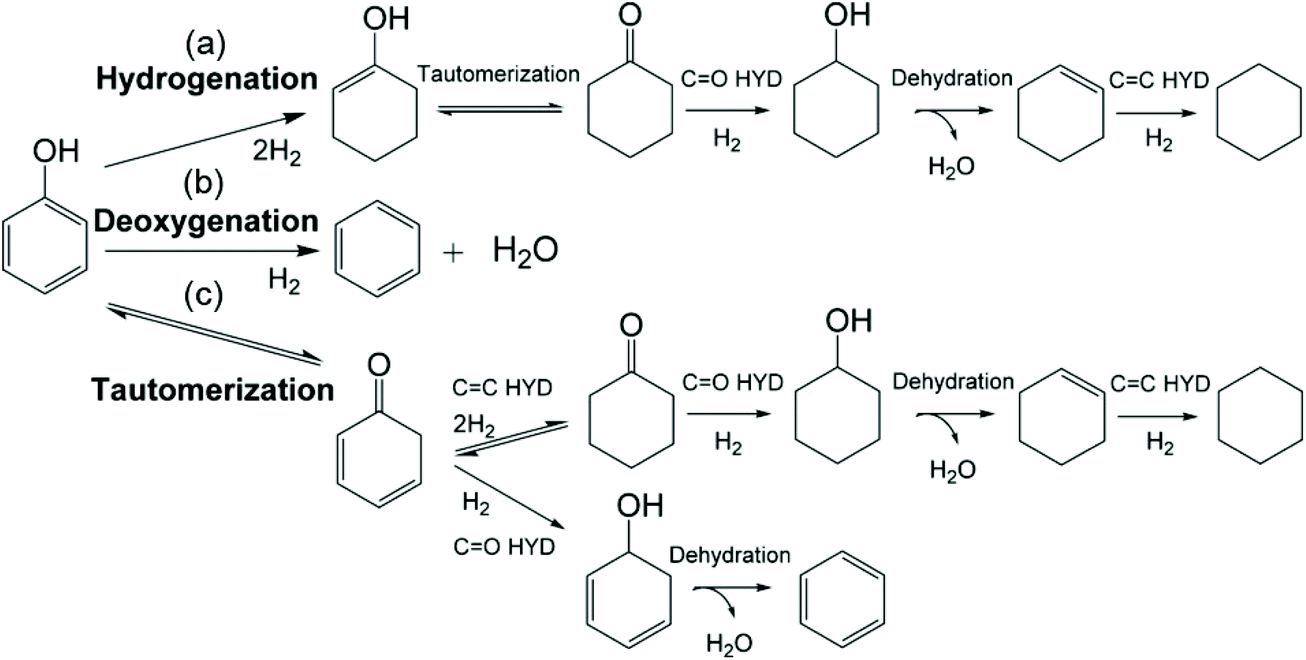
Hydrodeoxygenation of lignin-derived phenolics – a review on the active sites of supported metal catalysts - Green Chemistry (RSC Publishing) DOI:10.1039/D0GC02610G
![SOLVED: 22) The dehydration of secondary and tertiary akohols by reaction with POCh in pyridine is an example of: CH El process CH; B. an SN] process E2 process D: SN2 process SOLVED: 22) The dehydration of secondary and tertiary akohols by reaction with POCh in pyridine is an example of: CH El process CH; B. an SN] process E2 process D: SN2 process](https://cdn.numerade.com/ask_images/f54443461002458894aa1fe6f9dccbd4.jpg)
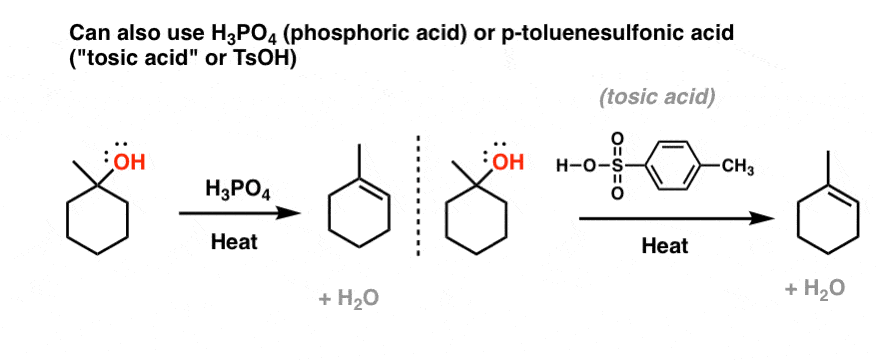
SOLVED: 22) The dehydration of secondary and tertiary akohols by reaction with POCh in pyridine is an example of: CH El process CH; B. an SN] process E2 process D: SN2 process

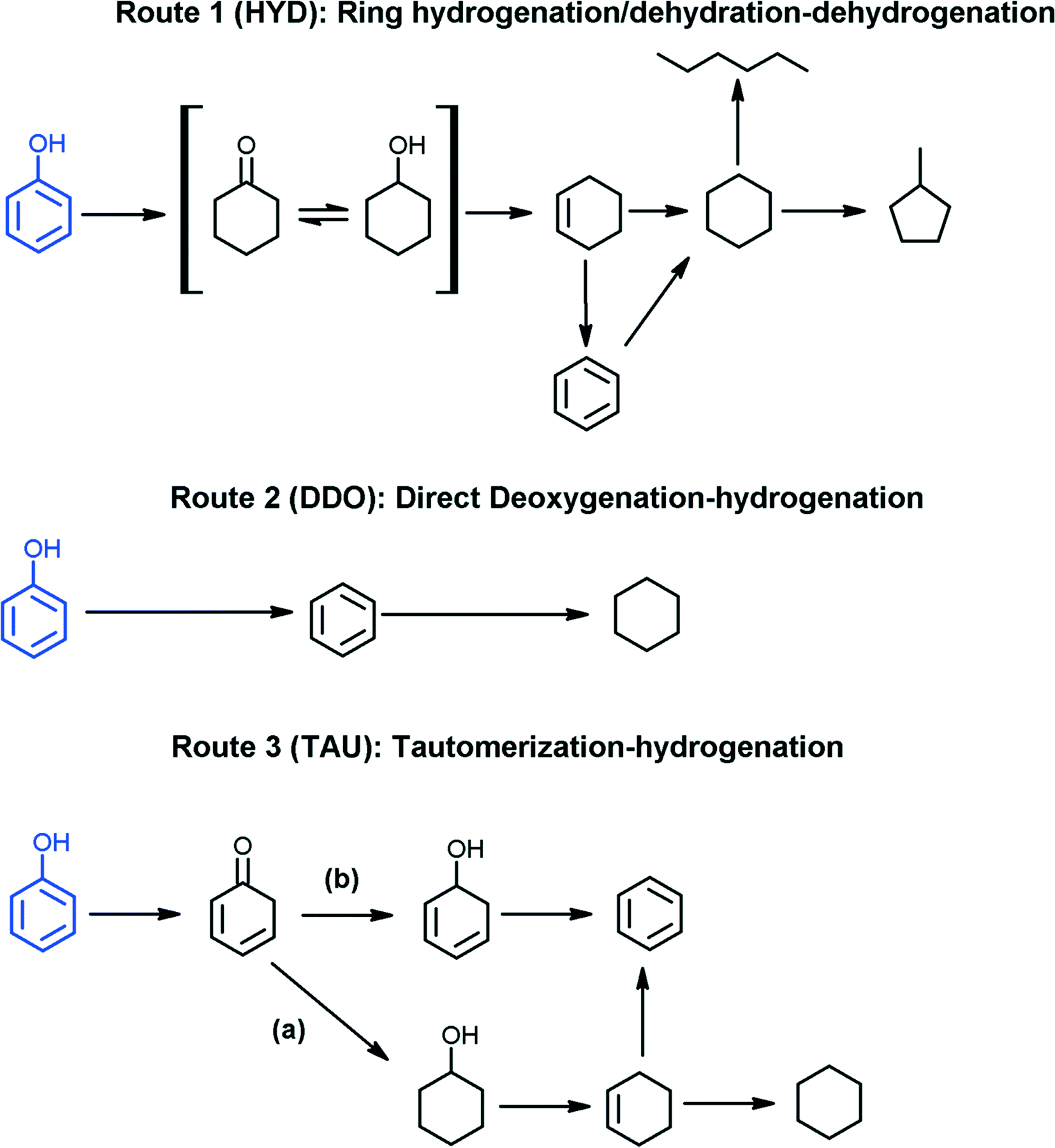
Mechanism of Dehydration of Phenols on Noble Metals via First-Principles Microkinetic Modeling | ACS Catalysis

Write the chemical reaction that occurs when phenol is mixed with water and name the product. | Homework.Study.com
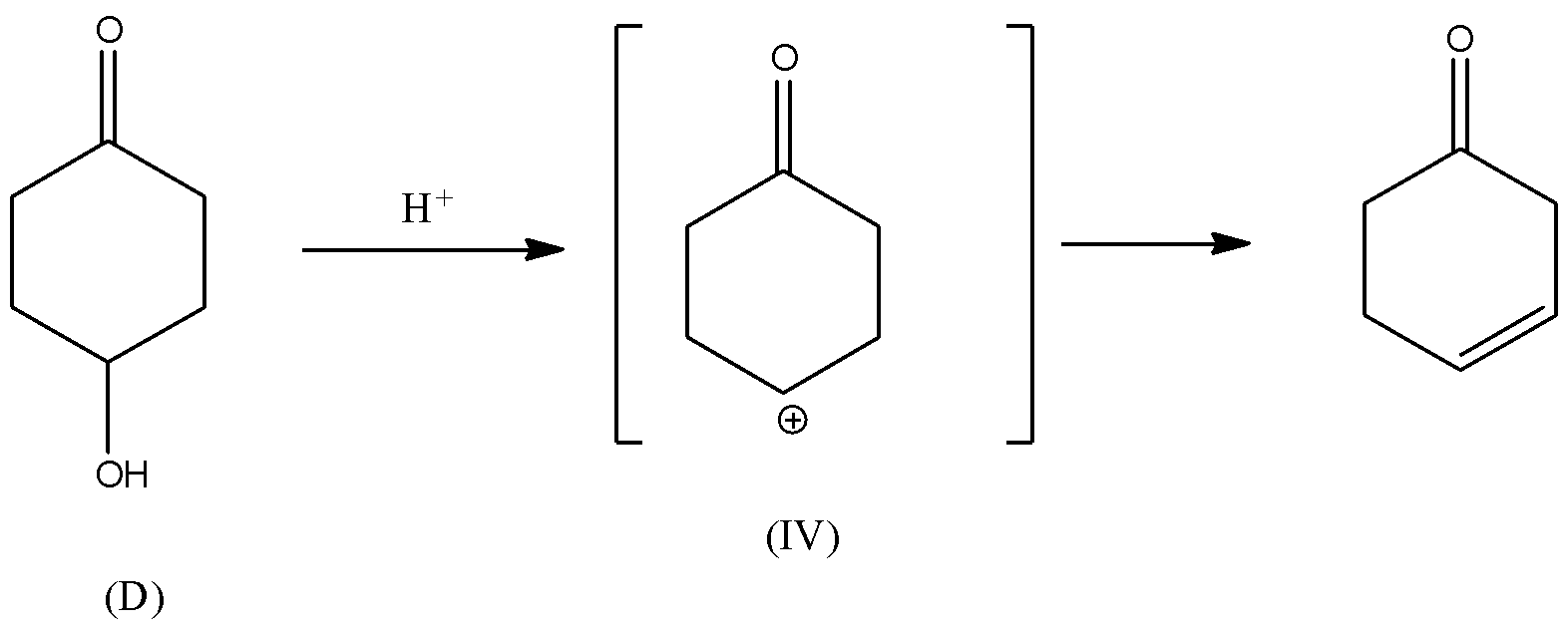
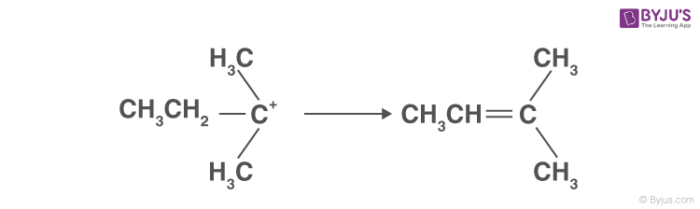
Which of the following alcohol will have the fastest rate of dehydration?A) \n \n \n \n \n B) \n \n \n \n \n C)\n \n \n \n \n D)\n \n \n \n \