
FDA Approves Foundation Medicine's FoundationOne CDx™, the First and Only Comprehensive Genomic Profiling Test for All Solid Tumors Incorporating Multiple Companion Diagnostics | Business Wire
FoundationOne®CDx Technical Information Foundation Medicine, Inc. 150 Second Street, Cambridge, MA 02141 Phone: 617.418.2200 I
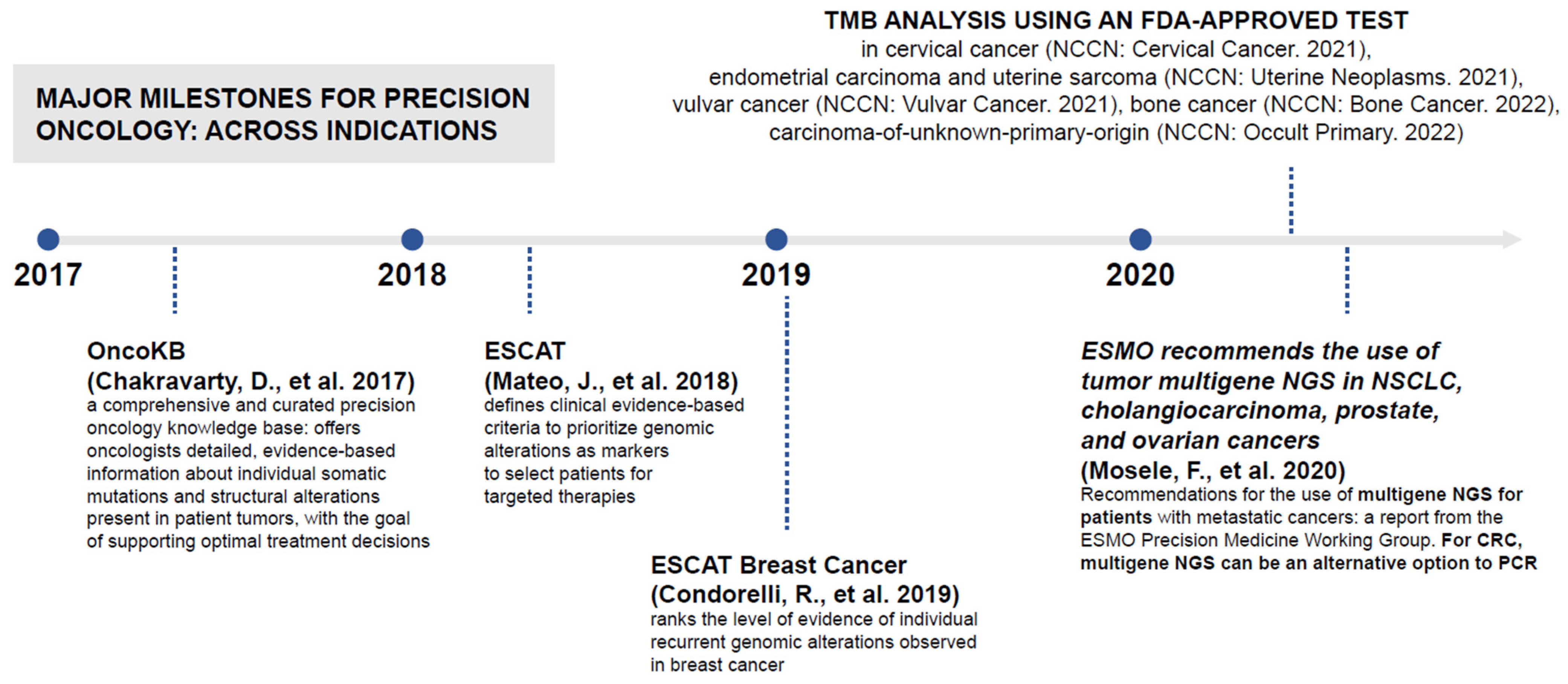
JPM | Free Full-Text | Identifying the Steps Required to Effectively Implement Next-Generation Sequencing in Oncology at a National Level in Europe
Clinical and analytical validation of FoundationOne®CDx, a comprehensive genomic profiling assay for solid tumors | PLOS ONE

Analytical Validation of a Hybrid Capture–Based Next-Generation Sequencing Clinical Assay for Genomic Profiling of Cell-Free Circulating Tumor DNA - ScienceDirect

FDA Approves Foundation Medicine's FoundationOne CDx™, the First and Only Comprehensive Genomic Profiling Test for All Solid Tumors Incorporating Multiple Companion Diagnostics | Business Wire
![Workflow for TMB assessment using the FoundationOne CDx assay [63].... | Download Scientific Diagram Workflow for TMB assessment using the FoundationOne CDx assay [63].... | Download Scientific Diagram](https://www.researchgate.net/publication/334081089/figure/fig2/AS:774610417090560@1561692923137/Workflow-for-TMB-assessment-using-the-FoundationOne-CDx-assay-63-ExAC-Exome.png)
Workflow for TMB assessment using the FoundationOne CDx assay [63].... | Download Scientific Diagram

When should we order a next generation sequencing test in a patient with cancer? - eClinicalMedicine

Concordance analysis of microsatellite instability status between polymerase chain reaction based testing and next generation sequencing for solid tumors | Scientific Reports

Next generation sequencing‐based gene panel tests for the management of solid tumors - Nagahashi - 2019 - Cancer Science - Wiley Online Library
Clinical and analytical validation of FoundationOne®CDx, a comprehensive genomic profiling assay for solid tumors | PLOS ONE

When should we order a next generation sequencing test in a patient with cancer? - eClinicalMedicine

Frontiers | The Impact of Foundation Medicine Testing on Cancer Patients: A Single Academic Centre Experience

FDA Approves Foundation Medicine's FoundationOne CDx™, the First and Only Comprehensive Genomic Profiling Test for All Solid Tumors Incorporating Multiple Companion Diagnostics | Business Wire
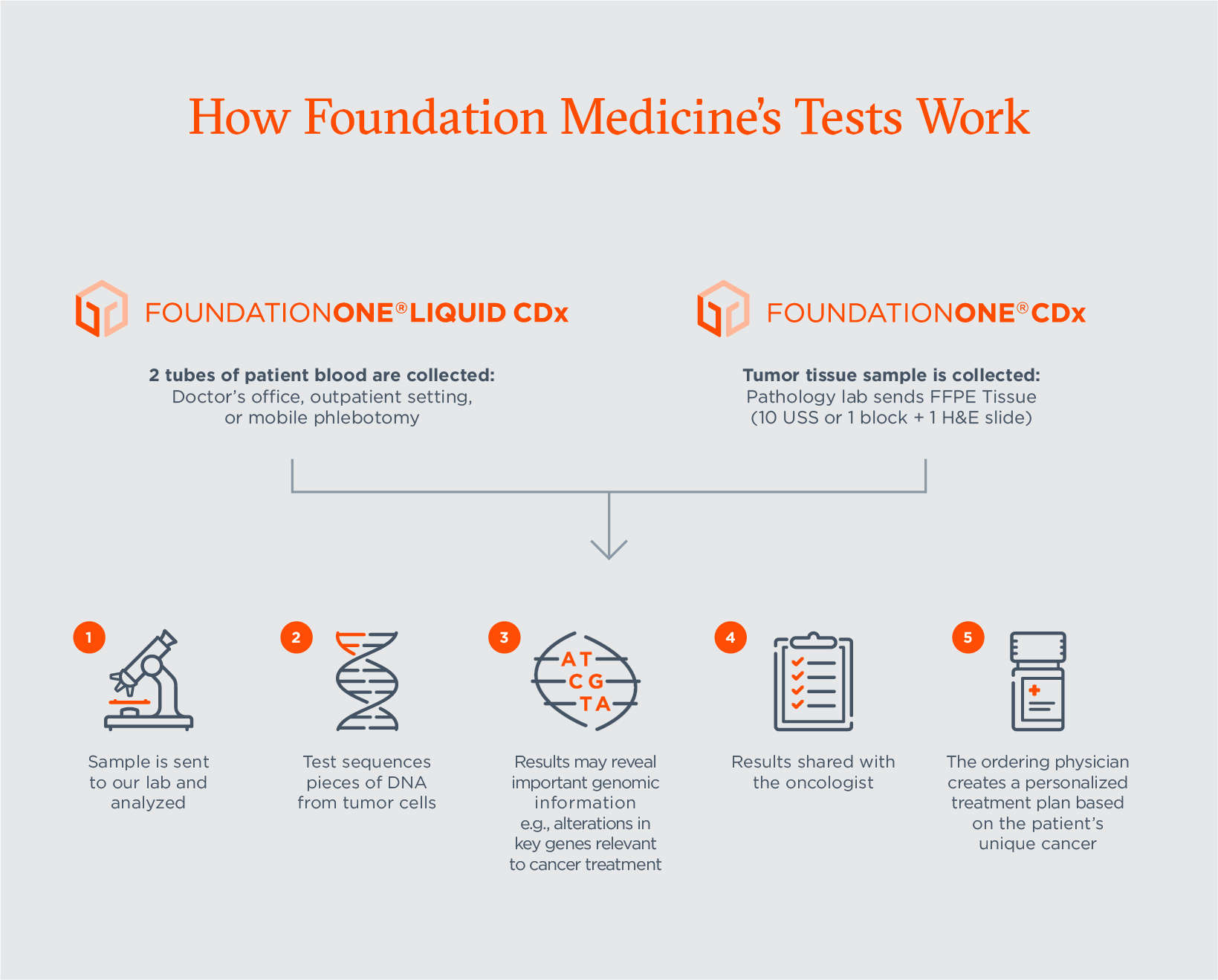
Companion Diagnostics Explained: Their Critical Role in Cancer Care and Our Latest Approvals | Foundation Medicine




