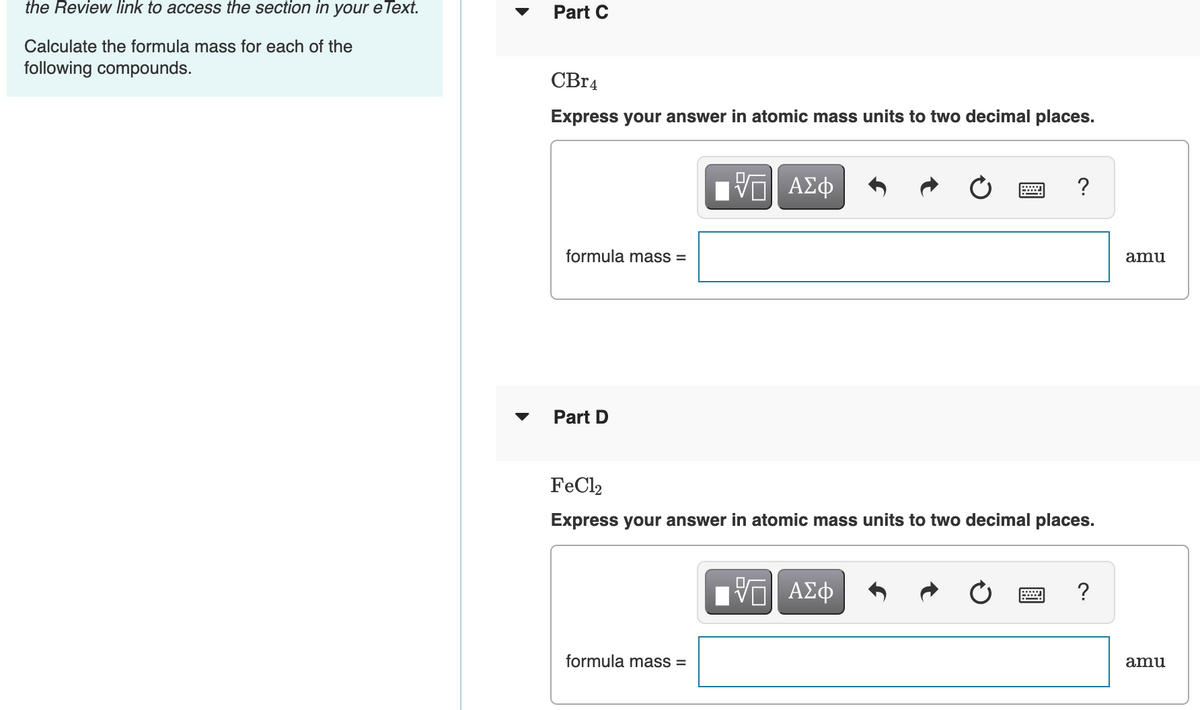
OneClass: Use the RefePences to access IMportant Values If leeded l8P tRiS qLEStIon Use the standard...

Cross-linked enzyme aggregates (CLEAs) of selected lipases: a procedure for the proper calculation of their recovered activity – topic of research paper in Biological sciences. Download scholarly article PDF and read for

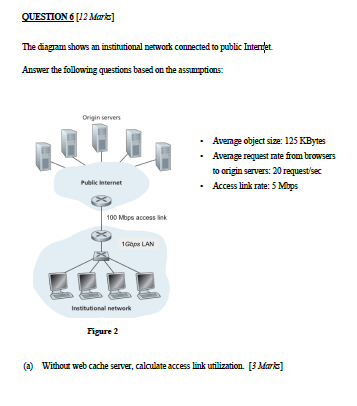
Application Layer 2-1 Chapter 2 Application Layer Computer Networking: A Top Down Approach 6 th edition Jim Kurose, Keith Ross Application Layer – Lecture. - ppt download

SOLVED: Use the References - access important ralues needed for this question Use the standard reduction potentials located in the 'Tables' linked above to calculate the standard free energy change in kJ

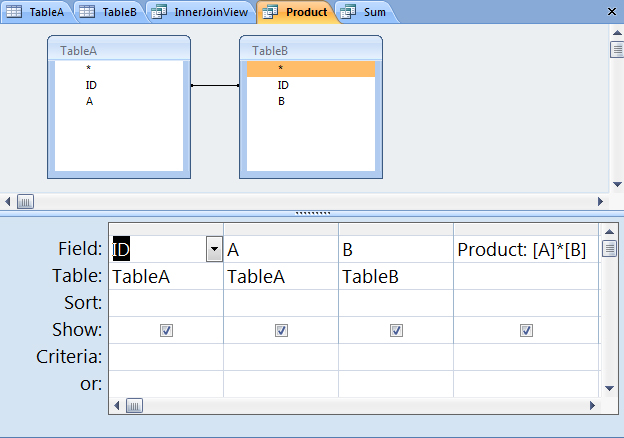
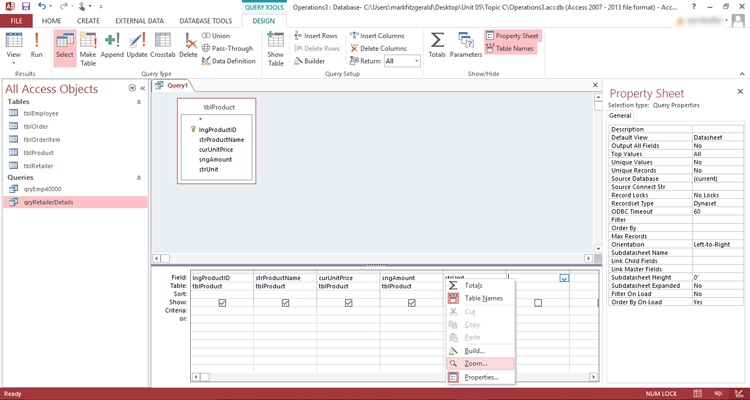
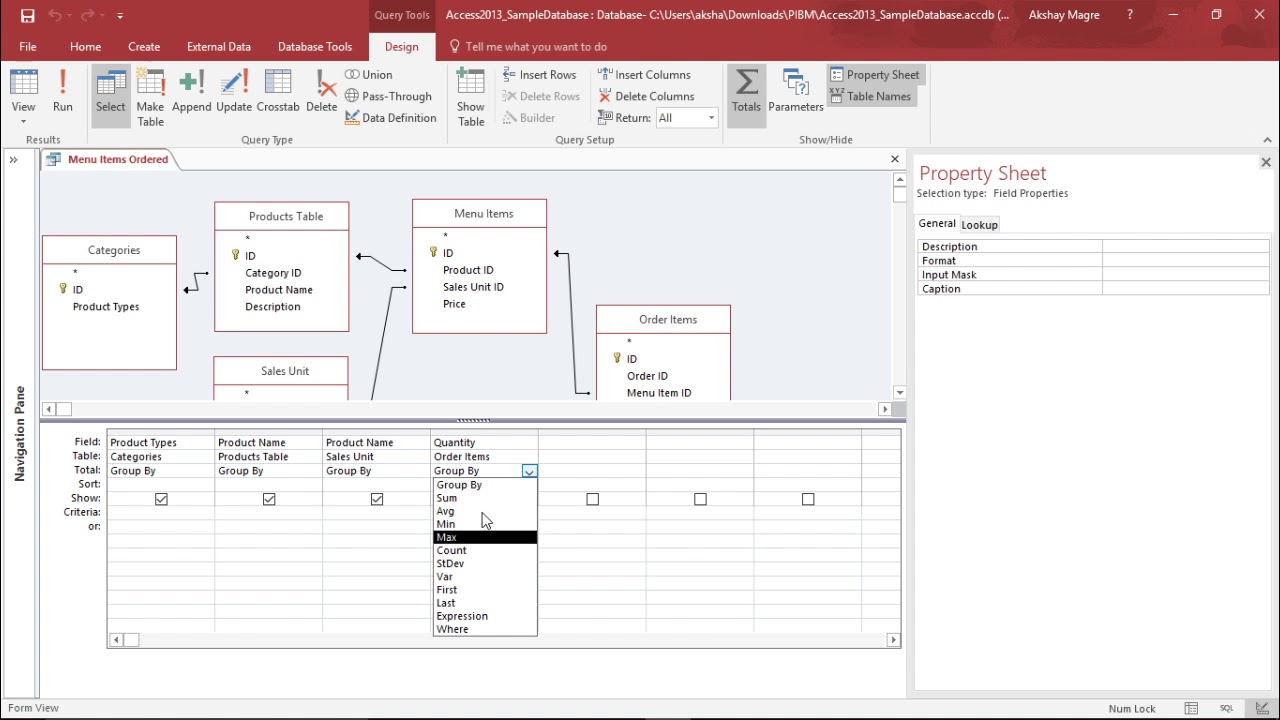
Calculated Fields in Microsoft Access - How to Perform Calculations in Queries & Form Footer Totals - YouTube

NIS SVG on Twitter: "Our e-Submit web application allows employers to submit contribution schedule information electronically. Calculations and error checking are automatically done. Visit the following link to access: https://t.co/NWpSsETBYY https://t ...
![SOLVED: [References] TUTOR Predicting Enthalpy of Reaction Using Bond Energies Use the References to access important values if needed for this question. Use average bond enthalpies (linked above) to calculate the enthalpy SOLVED: [References] TUTOR Predicting Enthalpy of Reaction Using Bond Energies Use the References to access important values if needed for this question. Use average bond enthalpies (linked above) to calculate the enthalpy](https://cdn.numerade.com/ask_images/a0fca3361e944bd58a91a904a33e3869.jpg)